Description
Lactic acid is a normal intermediate in the fermentation (oxidation, metabolism) of sugar. The concentrated form is used internally to prevent gastrointestinal fermentation. Lactic Acid, DL- is the racemic isomer of lactic acid, the biologically active isoform in humans. Lactic acid or lactate is produced during fermentation from pyruvate by lactate dehydrogenase. This reaction, in addition to producing lactic acid, also produces nicotinamide adenine dinucleotide (NAD) that is then used in glycolysis to produce energy source adenosine triphosphate. 2-hydroxypropanoic acid is a predicted metabolite generated by BioTransformer¹ that is produced by the metabolism of propanoic acid. It is generated by cyp2b6 and cyp2e1 enzymes via a hydroxylation-of-aliphatic-secondary-penultimate-carbon reaction. This hydroxylation-of-aliphatic-secondary-penultimate-carbon occurs in humans.
Storage Information: Hygroscopic. Keep tightly closed.
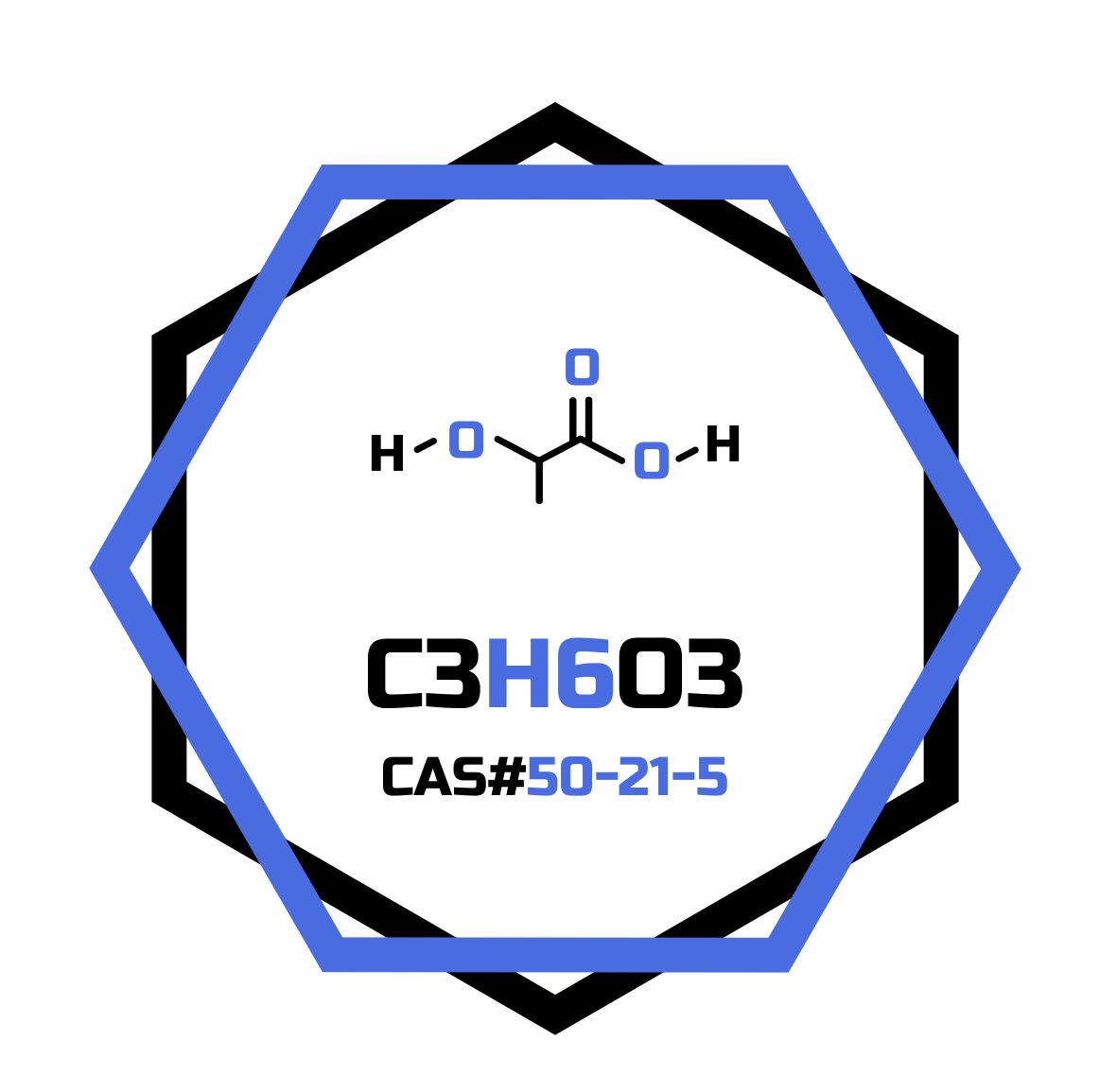
- Formula
- C3H6O3
- Molar mass
- 90.078 g/mol
- CAS Number
- 50-21-5
- Density
- 1.209 g/mL at 25 °C (lit.)
- Purity/Grade
- ~90%
- Apearance
- Liquid
- Initial boiling point and boiling range
- 122 °C (252 °F) at 20 hPa (15 mmHg)
- Flash point
- 113 °C (235 °F) – closed cup
Reviews
There are no reviews yet.